Complete Blood Morphology: A First Look at Fully Autonomous Digital Morphology
From the ADLM 2025 Conference | In-Booth Session
At ADLM 2025, we shared the first public look at Complete Blood Morphology (CBM) - a fully autonomous digital morphology analyzer designed to transform routine blood smear review. Two clinicians from Brigham and Women's Hospital joined us to discuss real-world workflow challenges and early performance data from the CBM pre-clinical study.
Watch the full session or read the key highlights below.
The Challenge: A Workforce Under Pressure
Dr. Daniel Dees, who oversees hematology operations at Brigham and Women’s Hospital – an 800-bed Level 1 trauma center and comprehensive cancer center – opened with a stark reality facing labs nationwide.
The math doesn’t add up:
“The US Bureau of Labor Statistics estimates approximately 2,500 jobs are going to be open per year for the next ten years, with approximately 5,000 graduates coming out of the programs. So we know that math doesn’t add up.”
Experience is walking out the door:
“An estimated 25 to 35% of our MLS workforce is going to reach retirement age within the next 3 to 5 years. This is really the one that keeps me up at night… Morphology is experience-based. How good you are at it directly correlates with how long you’ve done it.”
With volumes increasing 7–9% year over year at many institutions – well above the standard 3% – and manual differentials consuming significant technologist time, labs are stretched thin.
What Labs Actually Need: Better Resource Allocation
Dr. Dees shared what his team achieved by reducing their manual differential rate from 15% to 6% through workflow optimization – before CBM.
“I did not disregard any techs. I didn’t let any FTEs go. I was able to allocate my resources better.”
The results:
- Shifted night staff from five 8-hour shifts to four 10-hour shifts – improving work-life balance and retention
- Reduced overtime costs significantly
- Reallocated 2–4 FTEs to support integrated laboratory services across sites
- Brought stability to historically unstable shifts
CBM is designed to push that differential rate even lower – targeting a floor of approximately 7–10%.
The Trust Question: Can Automation Deliver?
Dr. Chris Hergott, a hematopathologist at Brigham and Women’s, addressed the question every lab professional asks about new technology:
“As a hematopathologist – and I think anyone in the hematology field – we sort of intrinsically understand that automation is going to become part of our lives. It’s an inevitability. The questions that remain are: What is the nature of that automation? Is it actually going to make my life easier? Can I trust it? We’re in a field where the error threshold is essentially zero.”
His answer after working with CBM in the pre-clinical study:
“For someone who is used to looking at glass, I find that to be very reassuring because you get the efficiency but you also get the verification.”
More Cells, Better Morphology
CBM analyzes significantly more cells than manual review – providing statistical rigor that wasn’t previously possible.
“You get a thousand white blood cells analyzed over 20 morphologic categories and axes of review, you get 10,000 red blood cells because you have the whole slide at your disposal, and you get five times more platelets. So that means more diagnostic sensitivity and that means more statistically rigorous results.”
Detecting What Manual Methods Miss
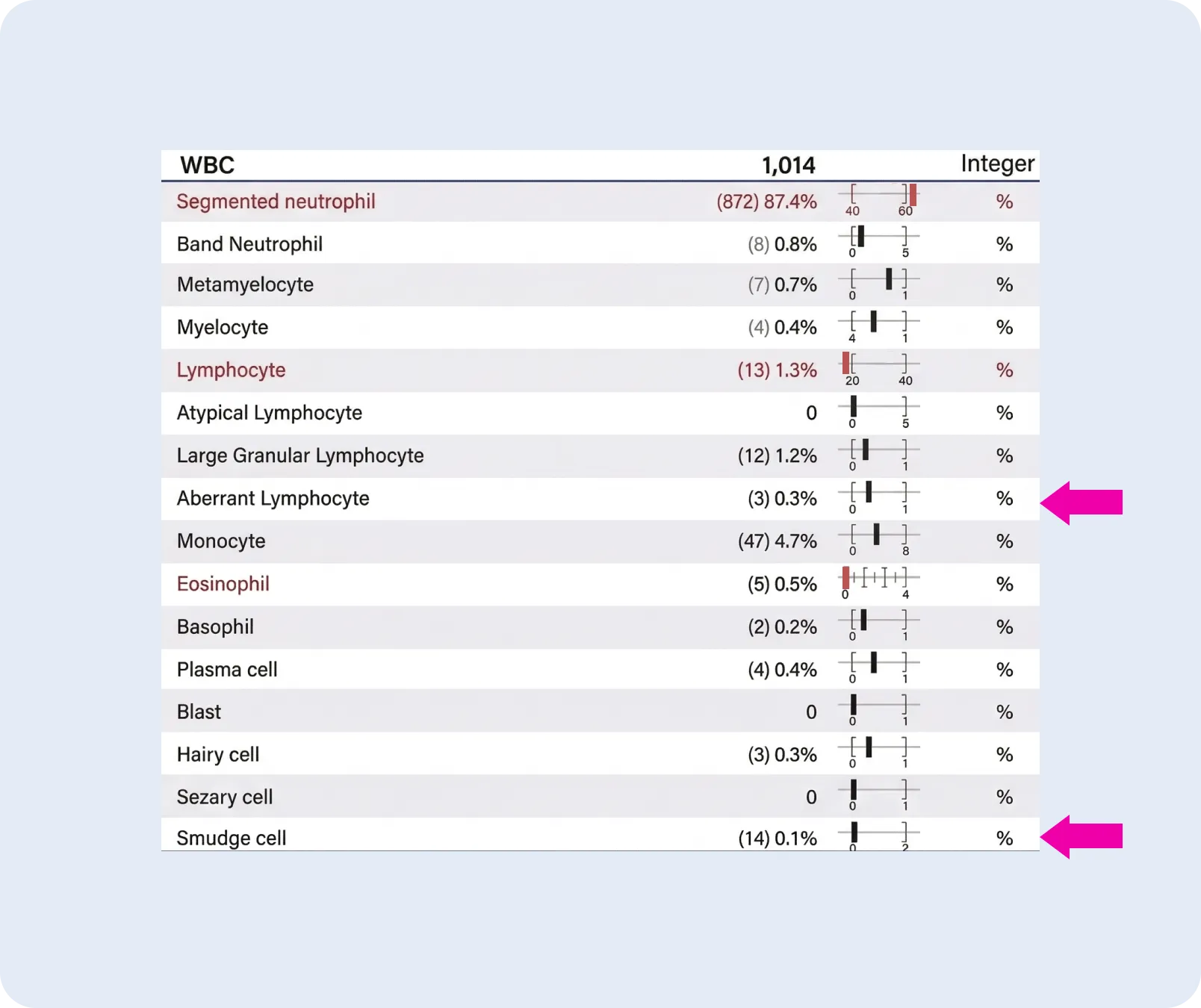
One of the most compelling use cases: identifying rare or low-burden findings that fall below manual sensitivity thresholds.
“When you have 200 cells, your diagnostic sensitivity is at best 0.5%. Anything that is relatively rare or low circulating burden, you’re not going to be reliably detecting.”
With CBM’s larger sample size, rare events show up – and show up reproducibly:
“In this example: 0.1% smudge cells, 1.2% LGL cells, and 0.3% aberrant lymphocytes with nuclear abnormalities that may raise suspicion for a low burden of a lymphoid neoplasm. Now with this information, you can make an informed decision about whether it’s worth sending for flow cytometry – as opposed to just blindly sending everybody, which is a suboptimal use of laboratory resources.”
Solving the Reproducibility Problem
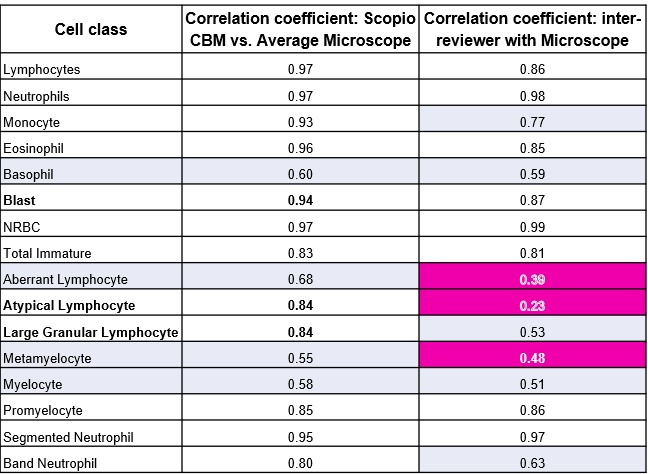
Lymphocyte classification has long been a challenge in manual morphology – even among experienced professionals.
“I can read a slide, you can read a slide – we’re going to get two different answers a lot of times.”
Dr. Hergott presented data showing human-to-human agreement on lymphocyte subtyping hovers in “coin-flip territory”:
“Digging in a little bit more on the lymphocyte data: 0.23, 0.53, 0.39 Pearson correlations between humans. That’s not really helpful for the patient. We’re doing the best we can. But you’re seeing doubling of the Pearson correlation with the software.”
This isn’t a criticism of technologists. It’s the nature of subjective morphological assessment. CBM introduces standardization where it wasn’t previously possible.
93% Full Automation
In validation testing, CBM demonstrated high throughput with minimal human intervention:
“We took 2,500 routine blood smears and said: let’s try to put these through the fully automated pathway and see what percentage has no eyes on it – from slide gets analyzed to into your LIS. That means no manual review, and even no computerized manual review. Doesn’t get flagged at all. 93% of blood smears go all the way through.”
The remaining 7% route to Scopio AI decision support, with the added benefit of Full-Field context for every flagged cell.
“Daniel mentioned a moment ago: through a considerable amount of excellent work and effort from the lab, the manual review rate went from about 15% to about 9%. That’s a huge achievement. But the floor here is 7%. And with further iterations and optimization in the lab, that will only get lower.”
The Bottom Line
“The idea is: not only is your data better, but it’s going to come at you faster, and you can trust it.”
What’s Next for CBM
Complete Blood Morphology is currently in pre-submission with the FDA. Scopio is conducting clinical studies with leading US institutions to validate performance across diverse patient populations.
Interested in learning more? Contact us to discuss how CBM could fit into your lab’s workflow.
Session Speakers

Recorded at ADLM 2025, Chicago
*CBM is currently in development and not yet available for diagnostic use in the United States. Performance data presented is from pre-clinical validation studies.